Biosimilars Causing A Stir In The U.S. Market, Are You Ready?
Barriers to biosimilar uptake in the U.S. temper optimistic predictions of accelerated biosimilars market growth.
Wondering why the opening of the U.S. market to biosimilars isn’t resulting in a collective cheer or groan from the biopharmaceuticals industry? Despite the FDA approval of the first biosimilar drug, Zarxio, in the U.S. earlier this month, the complex factors influencing the biologics market suggest that biosimilars will not overtake the market as rapidly as generics.
Biosimilars Disrupt Business For Biopharmaceutical Giants
Investors were unconcerned as the dip in Amgen’s stock after Zarxio’s approval rebounded a few days later after promising data was presented about their new biologic that lowers cholesterol.[i] The California biotech giant has also invested in their own biosimilars. Companies like Merck, Hospira and Sandoz, are aggressively pursuing biosimilars seeking rapid growth in the biosimilars market. With the Zarxio entry into the U.S., optimism is tempered by factors such as a stable of new biologics and biobetters (drugs engineered to be more effective than the original) in the pipeline, barriers in the U.S. market, and disappointing rates of biosimilar adoption observed in the European Union (EU). [ii],[iii]
Biosimilar Introduction In The U.S.
While chemically synthesized small molecule drugs have stiff competition from synthetic copies, or generics; biologics, or drugs produced by a biologic process, have been hard to imitate. Biosimilars, copies of biologics, entered the EU once their approval process launched in 2007. FDA rules for the approval of biosimilars have only recently been defined in the U.S. Amgen’s Neopogen brand, filgrastim, is used to prevent infection in immune-compromised cancer patients undergoing chemotherapy. Neopogen is the first of several biologics facing competition from a biosimilar as their U.S. patents expire.
Branded Biologics – Timeline For U.S. Patent Expiration[iv],[v]
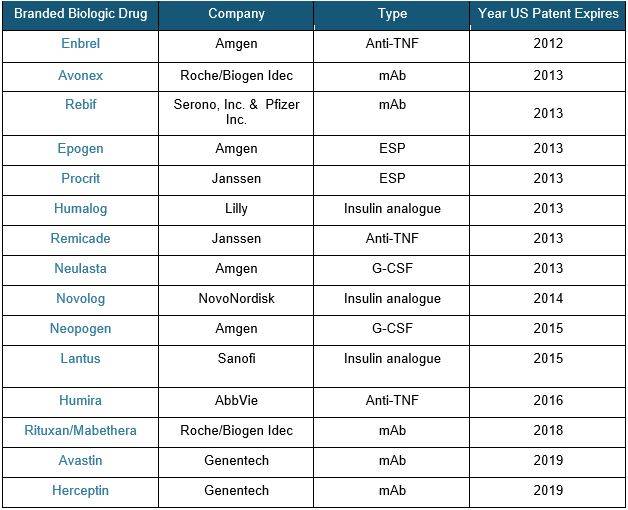
Once patents for these branded biologics expire, competition between generics, biobetters, and biosimilars expect to greatly increase within the next five years.
Market Growth Rate Defined By Payers, Physicians, And Patients
As the U.S. market opens, predictions for biosimilars market growth are cautious. The rate of EU physicians and patients embracing biosimilars necessary to fuel growth has been slower than expected. 3, 5 While U.S. payers, such as ExpressScripts, hail the arrival of biosimilars in the U.S. saying Zarxio could save the U.S. healthcare system $5.7 billion; prices for biosimilars in the EU have not been as deeply discounted as generics (25-30% vs. 50-80%).[vi], [vii]
A lack of familiarity may prevent physicians from suggesting biosimilar substitutions. Without deep price cuts, physicians and patients can weigh concerns about efficacy more heavily.3
Since U.S. pharmacists can’t automatically substitute Zarxio for Neupogen because it is therapeutically similar, not equivalent, conversion from the branded biologic may be slow. U.S. payers have no automatic incentives to substitute biosimilars. Moreover, adoption of biosimilar oncology drugs in the U.S. requires a channel change from provider-administered channels to specialty pharmacies, which could cause a lagging patient response. [viii]
Nonetheless, Zarxio is a good U.S. market entry choice, because the filgrastim biosimilar has achieved adoption in Europe at levels similar to a generic. In contrast, somatropin, used to treat a chronic condition in pediatric patients, has lagged in patient and physician support.[ix] Given the barriers to entry into the U.S. market, Zarxio (the Sandoz version of filgrastim) is more likely to pay-off.
Approvals Needed To Ignite Biosimilars Market Growth
The overall outlook for biopharma remains strong with the introduction of biosimilars. The opening of the U.S. market to biosimilars heralded by the Zarxio approval will take time to translate to adoption and cost savings for patients, payers and market growth for biosimilar manufacturers. Approval and acceptance of mAb and insulin analogue biosimilars in the EU, U.S. and emerging markets will define the rate of the biosimilars market expansion over the next few years.
By: Yolanda Fintschenko, PhD
References:
[i] Timmerman, L. “Amgen, Regeneron stocks go up as drugs bring cholesterol down,” Forbes. 16 March 2015, http://www.forbes.com/sites/luketimmerman/2015/03/16/amgen-regeneron-stocks-go-up-as-drugs-bring-cholesterol-down/.
[ii] Rickwood, S. and Di Biase, S. “Searching for Terra Firma in the Biosimilars and Non-Original Biologics Market Insights for the Coming Decade of Change,” IMS Health. ©2013.
[iii] McKinsey&Company, “Generating value in generics: Finding the next five years of growth,” © May 2013, Generating_value_in_generics_June 2013.pdf.
[iv] “Biologics, biosimilars, and generics. Preparing for next generation challenges,“ Analysis Group Health Care Consulting Bulletin. Fall/Winter 2010, http://www.analysisgroup.com/biologics_biosimilars_generics.aspx.
[v] Baxt, J. “When will biosimilars impact the US market?” BioSpace. 30 October 2014, http://www.biospace.com/News/when-will-biosimilars-impact-the-u-s-market/352139#sthash.9sLHS032.dpuf
[vi] King, S. “ViewPoints: Approval of the first US biosimilar – the key questions,” FirstWord Pharma. 8 March 2015, http://www.firstwordpharma.com/node/1267874#axzz3UgH5VSsJ
[vii] Kroll, D. “FDA approves first US biosimilar; hold your breath on cost savings,” Forbes. 6 March 2015, http://www.forbes.com/sites/davidkroll/2015/03/06/fda-approves-first-us-biosimilar-zarxio-by-sandoz/3/
[viii] Fein, A.J. “Zarxio: How channel dynamics will limit the first U.S. biosimilar,” Drug Channels. 10 March 2015, http://www.drugchannels.net/2015/03/zarxio-how-channel-dynamics-will-limit.html
[ix] Welch, A.R. “What’s next for the biosimilars market,” Outsourced Pharma.13 March 2015, http://www.outsourcedpharma.com/doc/what-s-next-for-the-biosimilars-market-0001
